What is Concept Design?
The concept design phase is an early stage of product development where the overall idea of a product takes shape.
During this phase, key aspects are explored, including:
- User experience and usability
- Product styling and aesthetics
- Production technologies
- Cost considerations
At this stage, the goal is not to go into full technical detail, but to transform an initial idea into several well-developed, visualized, and commercially viable concepts.
- In medical device design, this phase is especially critical. It involves:
- Ensuring that the concept aligns with user and regulatory requirements
- Evaluating potential solutions to a medical need
- Defining a clear design intent
From Idea to Concept: Main Steps and Deliverables

1. Moodboards
The concept design phase often begins with the creation of moodboards.
These are collections of images used to:
- Align with the client’s vision
- Define the design direction
- Explore styles, shapes, colors, and finishes
Moodboards help answer key questions:
- What should the product feel like?
- What visual language best represents the brand?
They may include:
- Competitor products
- Similar devices
- Inspirations from different industries
At this stage, the product requirements list (defined during the kickoff meeting) is also updated and shared with the client.
2. Market Research
Alongside moodboards, market research is conducted to understand the competitive landscape.
This includes:
- Analyzing competitors’ design choices
- Identifying trends in medical device design
- Mapping products using positioning boards
This step helps define:
- Design directions
- Differentiation opportunities
Additionally, the team may perform:
- User profiling
- Workflow analysis
These activities ensure a deeper understanding of user needs.
Deliverable: A presentation outlining the competitive landscape and updated product requirements.
3. Concept Development
After research and exploration, designers begin generating ideas through rapid sketching.
This phase focuses on:
- Exploring multiple design possibilities
- Testing functional and aesthetic solutions
- Identifying what works and what doesn’t

Key questions include: What features can be integrated? Which style best fits the target users?
From these explorations:
- The most promising ideas are selected
- Concepts are refined and developed further
Collaboration with engineering and production teams ensures:
- Technical feasibility
- Alignment with manufacturing constraints
At this stage, teams also define:
- Technical requirements
- Production technologies
- Structural details (e.g., enclosures, fixing points, IP ratings)
Output: One or more feasible product concepts.
4. Concept Development & Presentation
Once concepts are validated, they are developed into 3D CAD models.
These models provide:
- Accurate dimensions
- A realistic view of the product
- Early validation of manufacturability
Although not fully engineered, a design concept already consider:
- Production constraints
- Assembly feasibility
The goal is to present the final look and feel of the product.
Depending on the project, outputs may include:
- Detailed sketches
- 3D CAD models
- Photorealistic renderings
Renderings provide a realistic representation of the final device by simulating its colors, materials, and finishes (CMF).
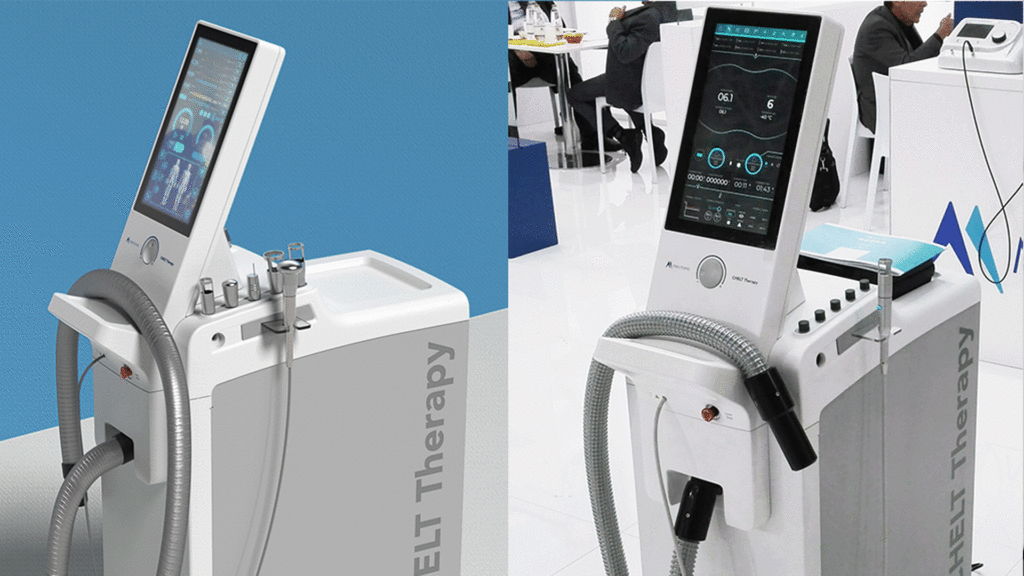
The final deliverable include a complete presentation with:
- Multiple render views of each concept
- Key features
- Design details
What Happens Next?
Together with the client, the best concept is selected. It is also possible to combine features from different concepts, creating a “mix and match” solution that integrates the most effective elements of each proposal.
The project then moves into the design refinement phase, where:
- Feedback is incorporated
- The concept is finalized
Once fully defined, the product is ready for engineering development.
Why This Phase Matters
Whether your medical device is:
- A disruptive innovation
- Or an improvement of an existing product
The concept design phase is the foundation for success.
At this stage, decisions made will influence:
- Usability
- Cost
- Manufacturability
- Market positioning
Creanova’s Approach
Creanova supports companies throughout this critical phase by combining:
- Creativity and innovation
- User-centered design
- Strong focus on production feasibility
From the very beginning, designs are developed with:
- Manufacturing constraints in mind
- Cost optimization strategies
The result is a producible and market-ready medical device concept.

Creanova integrates:
- Italian design excellence
- International experience
- Human-centered methodologies
This approach leads to intuitive and innovative healthcare solutions, recognized by leading design awards.
Creanova offers a full-service approach, covering:
- Concept design
- Product development
- Contract manufacturing
Ready to Bring Your Idea to Life? If you have a medical device idea and want to turn it into reality, now is the time to start.
Contact Creanova to take the first step in your product development journey.

