SmartOne OXI® – Combined Oximeter & Spirometer
The 16 years’ partnership between MIR and Creanova in design, product development and production, has brought to market another successful respiratory device.
Smart One OXI® was designed to self-manage Asthma, COPD, Lung Transplant care, Cystic fibrosis, enabling spirometry and oximetry. Thanks to the relevant app, the patient can share with a touch test results and his state of health with his doctor.

“Creanova consists of a great team of highly skilled specialists which offers effective design and a reliable manufacturing process. Being a strong turn-key partner is their winning excellence.”
Paolo Boschetti
CEO of MIR

Innovation and Compactness for Smart One OXI®
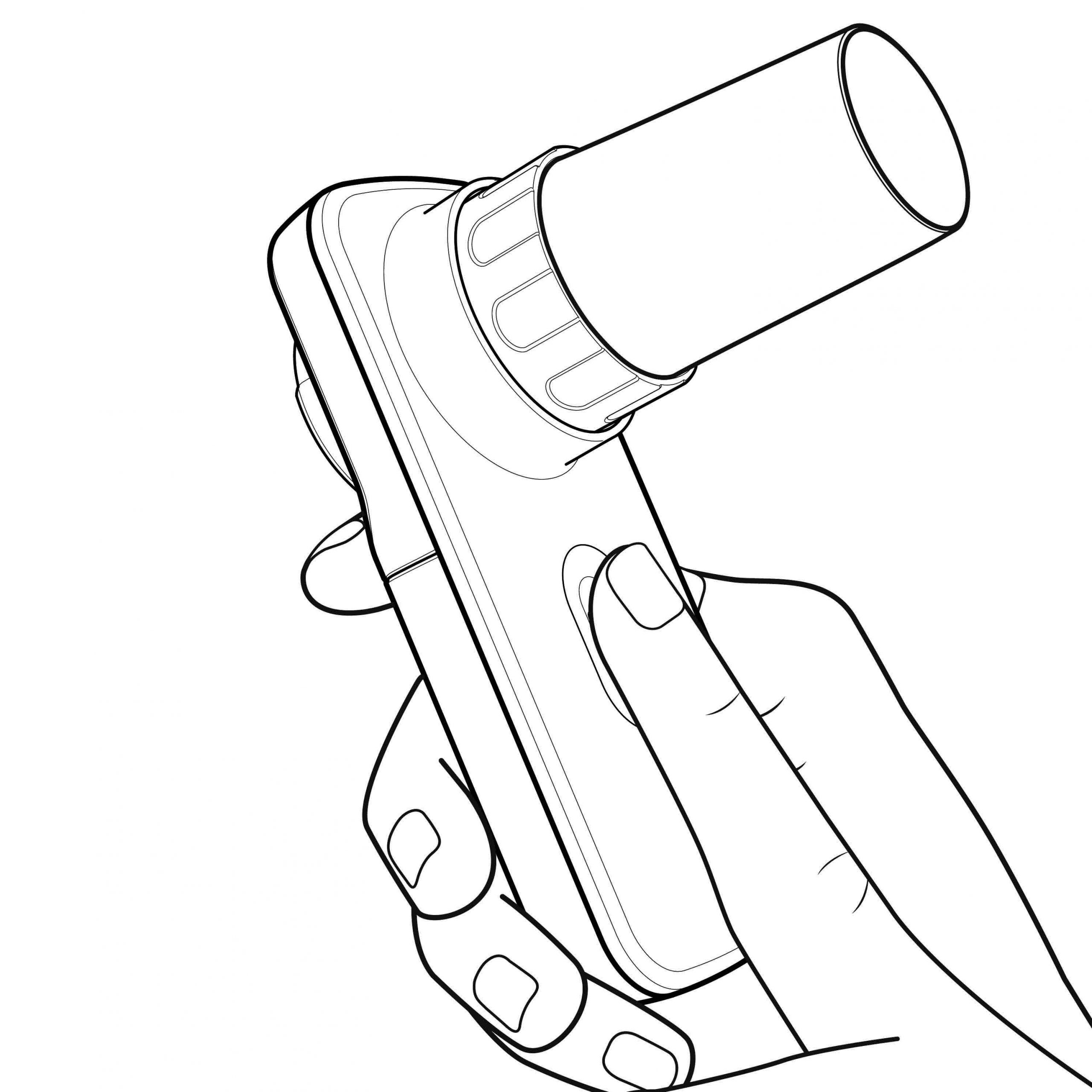
Due to the great customers feedback about the easy-to use, ergonomics and compactness of Smart One, MIR asked Creanova to review the well-known design and craft its new respiratory device.
Creanova’s designers updated the Smart One design to meet the new respiratory device requirements: a small opening to place the oximetry slide and a tiny thickening to make room for oximetry technology. The spirometer-oximeter was completed by the relative package and user manual.

New Layout Focused on Space Optimization
The main challenge in engineering phase was to maintain the successful size and shape of Smart One’s housings while include the oximetry PCB.
The layout of internal components was totally reviewed and the oximetry test through a small sensor on the top cover was enabled. These enhancements place the Smart One OXI® one step ahead of competitors, avoiding an additional accessory (such as the finger clip) and thus optimizing the user experience.
Rapid Prototyping
The volumetric analysis was carried out on a mock up by rapid prototyping.
Functional prototypes were made by CNC and part of these, equipped with an SD, were used to test the device.


Injection Molding, Assembly and Packaging
Production start-up team managed the mold making and injection molding of the plastic housings.
The top cover of the oximeter needs three over-moldings: main cover, the oximetry slide in clear plastics and the gasket.
The assembly was performed at Creanova’s production site, certified with ISO 13485.
Explore
How
We Work
Discover how our milestone-based approach reduces risk, ensures regulatory readiness, and accelerates your path to market.